Lupron Guinea Pigs: Two Decades of Experimentation on Autistic Children (Part One)
We take an in-depth look at Lupron, touted in the noughties as a quack 'cure' for autism, embraced later by doctors as 'gender affirming care'... but still somehow used primarily on autistic children.
In the mid-1990s, Dutch clinicians used the case of a sixteen-year-old girl, who was experimentally given Gonadotropin-Releasing Hormone agonist (GnRHa) triptorelin at the age of thirteen by her endocrinologist, to create a new protocol for administering hormonal treatments to children experiencing gender dysphoria before the normal age of consent. The ‘Dutch Protocol’ as it came to be known, was introduced to the British public consciousness shortly after when three million people in the UK, watched Channel 4’s 1996 documentary The Decision: The Wrong Body, in which British gender dysphoric young people went to Holland to meet peers who had received hormonal gender treatments as children. Stephen Whittle (a woman who claims a trans identity) who had set up Press for Change in 1992, to campaign on transgender issues in the media, began to push for young people in the UK to have access to these ‘puberty blockers’. Whittle had a long way to go, however; the UK’s biggest gender clinic, the Tavistock, would not begin to experiment with giving such treatments to children until 2011.
Meanwhile in the US, TAP Pharmaceuticals – a short-lived joint marketing venture between Japanese Pharmaceutical Company, Takeda, and Illinois-based Abbott Laboratories – was marketing its own GnRHa: Leuprolide. Patented in 1973, approved for medical use in the US in 1985 and better known by its brand name, Lupron, leuprolide was approved for use in cases of prostate cancer but by the mid-noughties it was also being used, off-label and unapproved by the US Food and Drugs Administration, as a ‘cure’ for autism. In 2004, father and son team, Mark and David Geier, filed a patent for their ‘Lupron Protocol’, which involved administering Lupron to autistic children along with chelation drugs (which strip heavy metals from the blood, the use of which has been fatal in at least one case, that of five-year-old Abubakar Nadama), asserting that the autism was caused by mercury poisoning, in turn caused by thimerosal in childhood vaccines. Despite being widely discredited, the Geiers (who also had a history of testifying for litigants in vaccine-injury cases) and their theories gained a following and they would soon have licences to practice in California, Florida, Hawaii, Illinois, Indiana, Kentucky, Missouri, New Jersey, Virginia and Washington as well as in their native Maryland.
In 2005, the Geiers were invited to speak about their ‘protocol’ at the anti-vaccine Autism One conference in Chicago, Illinois. A few years later, in 2008, the Geiers met obstetrician and “natural birth” advocate, Dr. Mayer Eisenstein, in the same city at the Health Freedom Expo and worked with him to set up his Homefirst Autism Recovery Clinic in Rolling Meadows in suburban Chicago. Over the next half decade, the Geiers, along with Dr. Eisenstein their other acolytes/business-partners, administered Lupron injections to autistic children across the US. In 2009, Geier Senior claimed he had treated up to 600 children and a handful of adults with the drug himself.
Lupron did not cure anyone of autism but it did make purveyors of the Geiers’ protocol and TAP Pharmaceuticals a lot of money – Dr. Eisenstein reputedly charged $5 - 6,000 per month for the injections, which, in order to prevent puberty, had to be administered indefinitely, making patients-for-life. Lupron’s marketing team seemed to believe the Geiers’ protocol showed sales potential and in September 2005, mere weeks after chelation therapy caused Abubakar Nadama’s death, TAP Pharmaceuticals, Abbott Endocrine and Missouri-based Lake Products LLC (makers of key ingredient of the Lupron injection, Sodium Chloride) supported the Geiers’ Lupron Protocol patent, Methods of Treating Disorders Having a Component of Mercury Toxicity. The protocol claimed mercury caused autism and that Lupron and chelation could remove mercury from children’s brains. TAP’s marketing team was well acquainted with aggressive marketing strategies; in 2001, its Lupron-marketing techniques got the company embroiled in one of the biggest pharmaceutical lawsuits of all time and it was sued by US government programmes Medicare and Medicaid for $875 million for overcharging them for the drug; incredibly, just four years later, TAP was considering marketing Lupron as a ‘cure’ for autism based on the Geiers’ junk science. Despite the initial support, however, TAP and co. dropped out and by 2008, Abbott Labs and Takeda had disbanded TAP Pharmaceuticals altogether.
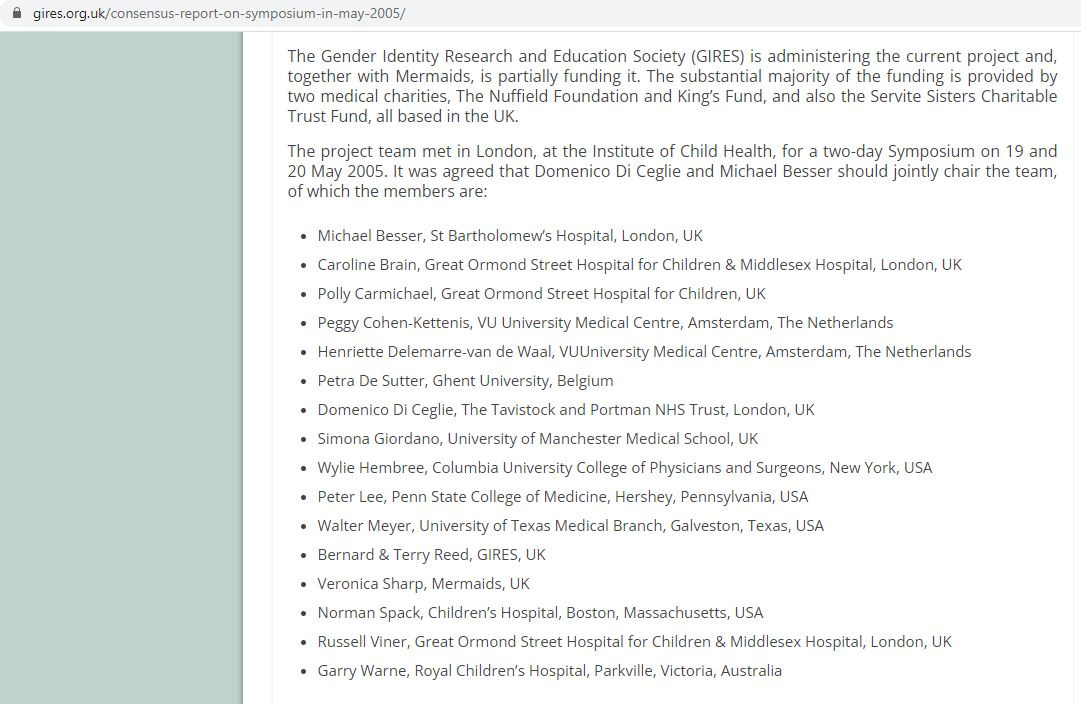
While this was going on, across the pond in the UK, two organisations – the Gender Identity Research and Education Society (GIRES) and Mermaids – had been founded and begun to campaign for hormone treatments to be available on the NHS to children identifying as transgender. In 2005, GIRES organized a symposium in London to develop “guidelines for endocrinological intervention” with Mermaids and other medical charities providing additional funding. The symposium was attended by the creators of the Dutch protocol, as well as Domenico Di Ceglie, Director of the Gender Identity Development Service (GIDS) and Great Ormond Street Hospital’s Polly Carmichael and Russell Viner, who would eventually begin hormonal treatment experiments on children at the Tavistock in 2011. Also in attendance was the American paediatric endocrinologist, Dr. Norman Spack.
In the Takeda/Abbott Laboratories 2008 settlement, Abbott Labs hung on to the rights to Lupron, which in 2007 had sales of $600 million and a patent which would not expire until 2015. In California, in the same year TAP Pharmaceuticals was making $600 million in Lupron sales, Sylvia Joanna Vazquez was convicted on horrific child neglect charges. Riffing on the extreme and dehumanising techniques she had learned from Nancy Thomas Parenting classes (and a book given to her by her social worker), she kept three of her four adopted children in cages with buckets for toilets and fed them a restricted diet of bread, water and peanut butter. To the twelve-year-old girl (the one not kept in a cage) she gave shots of Lupron, curtailing her physical development. The case set a precedent, arguably defining medically unnecessary administration of puberty-blocking hormone treatments as felony child abuse. Nevertheless, that same year, Dr. Spack opened his gender identity clinic at the Boston Children’s Hospital – the first in the US set up to treat children – where he began to administer Lupron as a puberty blocker. He assured parents of trans-identified children that Lupron injections ‘bought them time’ while they considered irreversible ‘gender affirming’ treatments of cross-sex hormones and surgeries.
One of Dr. Spack’s first patients was Jackie, son of Susie Green (and soon to become CEO of the charity Mermaids). Jackie was taken to Dr. Spack’s clinic for puberty blockers from 2007, when he was twelve years old. Around that time, in the UK, Mermaids gave a presentation to parents about transgender medical tourism and Dr. Spack saw several more British children for treatment at his clinic. Dr. Spack was likely to have been the person who advised Green about plans in Thailand to raise the legal age for ‘sex-change’ genital surgery from sixteen to eighteen. She booked Jackie into a Thai hospital for penile inversion surgery for his sixteenth birthday, squeaking in just before the age of consent was raised.
In part two, we explore Lupron’s side-effects, the explosion of child gender dysphoria referrals and TAP Pharmaceuticals/Abbot Laboratories’ Lupron sales techniques.